Cutaneous Mycoses
These are superficial fungal infections of the skin, hair or nails.
No living tissue is invaded, however a variety of pathological changes occur in the host because of the presence of the infectious agent and its metabolic products.
| Disease | Causative organisms | Incidence |
|---|---|---|
| Dermatophytosis Ringworm of the scalp, glabrous skin and nails. |
Dermatophytes (Arthroderma, Lophophyton, Microsporum, Nannizzia, Trichophyton, Epidermophyton) |
Common |
| Candidiasis of skin, mucous membranes and nails. |
Candida, Debaryomyces, Kluyveromyces, Meyerozyma, Pichia, etc. |
Common |
| Dermatomycosis | Non-dermatophyte moulds Neoscytalidium, Scopulariopsis |
Uncommon |
Mycoses descriptions
-
Dermatophytosis - Ringworm or Tinea
Dermatophytosis (tinea or ringworm) of the scalp, glabrous skin, and nails is caused by a closely related group of fungi known as dermatophytes which have the ability to utilise keratin as a nutrient source, i.e. they have a unique enzymatic capacity [keratinase].
The disease process in dermatophytosis is unique for two reasons: Firstly, no living tissue is invaded the keratinised stratum corneum is simply colonised. However, the presence of the fungus and its metabolic products usually induces an allergic and inflammatory eczematous response in the host.
The type and severity of the host response is often related to the species and strain of dermatophyte causing the infection. Secondly, the dermatophytes are the only fungi that have evolved a dependency on human or animal infection for the survival and dissemination of their species.
Clinical manifestations:
The common anthropophilic species are primarily parasitic on man. They are unable to colonise other animals and they have no other environmental sources. On the other hand, geophilic species normally inhabit the soil where they are believed to decompose keratinaceous debris.
Some species may cause infections in animals and man following contact with soil. Zoophilic species are primarily parasitic on animals and infections may be transmitted to humans following contact with the animal host.
Zoophilic infections usually elicit a strong host response and on the skin where contact with the infective animal has occurred ie arms, legs, body or face.
Ecology of common dermatophyte species. Species Natural habitat Incidence Epidermophyton floccosum Humans Common Trichophyton rubrum Humans Very Common Trichophyton interdigitale Humans Very Common Trichophyton tonsurans Humans Common Trichophyton violaceum Humans Less Common Trichophyton concentricum Humans Rare* Trichophyton schoenleinii Humans Rare* Trichophyton soudanense Humans Less Common* Microsporum audouinii Humans Less Common* Microsporum ferrugineum Humans Less Common* Trichophyton mentagrophytes Mice, rodents Common Trichophyton equinum Horses Less Common Trichophyton eriotrephon Hedgehogs Rare* Trichophyton verrucosum Cattle Less Common Microsporum canis Cats Common Nannizzia gypsea Soil Common Nannizzia nana Soil/Pigs Rare* Nannizzia fulva Soil Rare Nannizzia persicolor Voles and bats Rare* Lophophyton cookei Soil Rare* Lophophyton gallinae Chicken and other fowl Rare* * Geographically restricted. 
Tinea is transmitted via the feet by desquamated skin scales in substrates like carpet and matting.
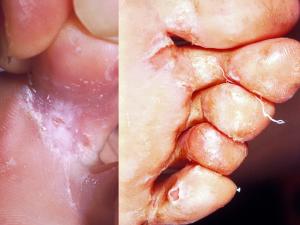
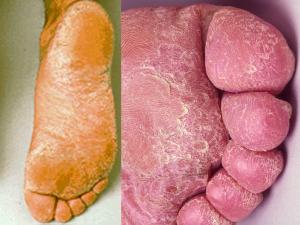
Tinea pedis:
Infections by anthropophilic dermatophytes are usually caused by the shedding of skin scales containing viable infectious hyphal elements [arthroconidia] of the fungus. Desquamated skin scales may remain infectious in the environment for months or years. Therefore transmission may take place by indirect contact long after the infective debris has been shed.
Substrates like carpet and matting that hold skin scales make excellent vectors. Thus, transmission of dermatophytes like Trichophyton rubrum, T. interdigitale and Epidermophyton floccosum is usually via the feet. In this site infections are often chronic and may remain subclinical for many years only to become apparent when spread to another site, usually the groin or skin.
It is important to recognise that the toe web spaces are the major reservoir on the human body for these fungi and therefore it is not practical to treat infections at other sites without concomitant treatment of the toe web spaces. This is essential if a "cure" is to be achieved. It should also be recognised that individuals with chronic or subclinical toe web infections are carriers and represent a public health risk to the general population, in that they are constantly shedding infectious skin scales.
Click images below to expand:
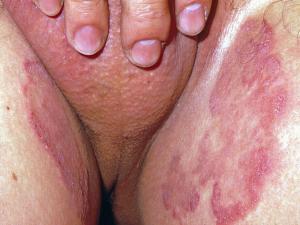
Tinea cruris:
Tinea cruris refers to dermatophytosis of the proximal medial thighs, preum and buttocks. It occurs more commonly in males and is usually due to spread of the fungus from the feet. Thus the usual causative agents are T. rubrum, T. interdigitale and E. floccosum.
Click images below to expand:
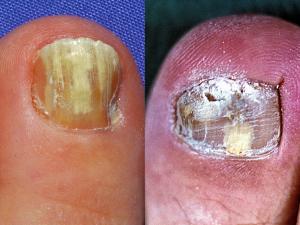
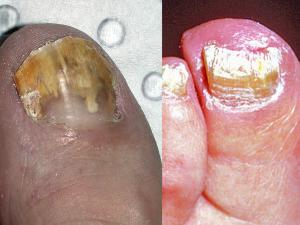
Tinea unguium (dermatophyte onychomycosis):
Trichophyton rubrum and T. interdigitale are the dominant dermatophyte species involved. In countries like Australia, UK and USA the incidence of dermatophyte onychomycosis has been estimated to be about 3% of the population, increasing up to 5% in the elderly, with some subgroups such as miners, servicemen and sportsmen etc having an incidence of up to 20% due to the use of communal showers and changing rooms.
It is important to stress that only 50% of dystrophic nails have a fungal aetiology, therefore it is essential to establish a correct laboratory diagnosis by either microscopy and/or culture, before treating a patient with a systemic antifungal agent.
Click images below to expand:
Dermatophyte onychomycosis may be classified into two main types; (1) superficial white onychomycosis in which invasion is restricted to patches or pits on the surface of the nail; and (2) invasive, subungual dermatophytosis in which the lateral, distal or proximal edges of the nail are first involved, followed by establishment of the infection beneath the nail plate. Distal subungual onychomycosis is the most common form of dermatophyte onychomycosis. The fungus invades the distal nail bed causing hyperkeratosis of the nail bed with eventual onycholysis, and thickening of the nail plate.
As the name suggests, lateral subungual onychomycosis begins at the lateral edge of the nail and often spreads to involve the entire nail bed and nail plate. In proximal subungual onychomycosis, the fungus invades under the cuticle and infects the proximal rather than the distal nail bed causing yellowish-white spots which slowly invade the lunula and then the nail plate.
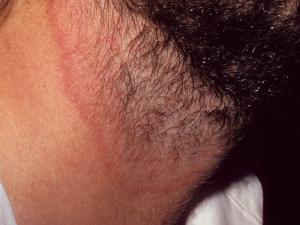
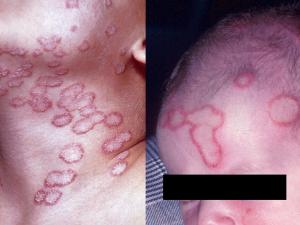
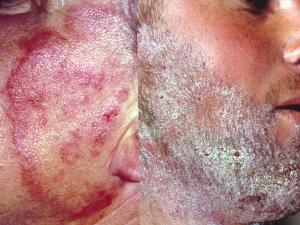
Tinea corporis:
Tinea corporis refers to dermatophytosis of the glabrous skin and may be caused by anthrophophilic species such as T. rubrum usually by spread from another body site or by geophilic and zoophilic species such as M. gypseum and M. canis following contact with either contaminated soil or an animal host.
Click images below to expand:
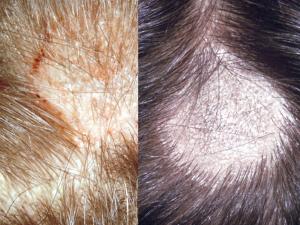
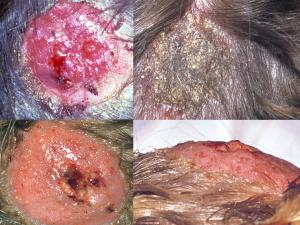
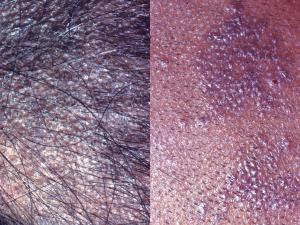
Tinea capitis:
Tinea capitis refers to dermatophytosis of the scalp. Three types of in vivo hair invasion are recognised:
Ectothrix invasion is characterised by the development of arthroconidia on the outside of the hair shaft. The cuticle of the hair is destroyed and infected hairs usually fluoresce a bright greenish yellow colour under Wood's ultraviolet light. Common agents include Microsporum canis, Nannizzia.gypsea, Trichophyton equinum and T. verrucosum.
Endothrix hair invasion is characterised by the development of arthroconidia within the hair shaft only. The cuticle of the hair remains intact and infected hairs do not fluoresce under Wood's ultraviolet light. All endothrix producing agents are anthropophilic eg Trichophyton tonsurans and T. violaceum.
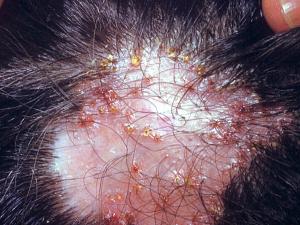
Favus usually caused by Trichophyton schoenleinii, produces favus-like crusts or scutula and corresponding hair loss.
Click images below to expand:
Laboratory diagnosis:
Clinical material:

Black collection card showing (a) a suitable amount of nail material for a good sample and (b) an inadequate specimen.
Skin Scrapings, nail scrapings and epilated hairs. For a laboratory diagnosis, clinicians should be aware of the need to generate an adequate amount of suitable clinical material. Unfortunately many specimens submitted are either of an inadequate amount or are not appropriate to make a definitive diagnosis. The laboratory needs enough specimen to perform both microscopy and culture. Routine turn around times for direct microscopy should be less than 24 hours, however culture may take several weeks.
In patients with suspected dermatophytosis of skin [tinea or ringworm] any ointments or other local applications present should first be removed with an alcowipe. Using a blunt scalpel, tweezers, or a bone curette, firmly scrape the lesion, particularly at the advancing border. In cases of vesicular tinea pedis, the tops of any fresh vesicles should be removed as the fungus is often plentiful in the roof of the vesicle.
In patients with suspected dermatophytosis of nails [onychomycosis] the nail should be pared and scraped using a blunt scalpel until the crumbling white degenerating portion is reached. Any white keratin debris beneath the free edge of the nail should also be collected.
Skin and nail specimens may be scraped directly onto special black cards which make it easier to see how much material has been collected and provide ideal conditions for transportation to the laboratory.
It must be stressed that up to 30% of suspicious material collected from nail specimens may be negative by either direct microscopy or culture. A positive microscopy result showing fungal hyphae and/or arthroconidia is generally sufficient for the diagnosis of dermatophytosis, but gives no indication as to the species of fungus involved. Culture is often more reliable and permits the species of fungus involved to be accurately identified. Repeat collections should always be considered in cases of suspected dermatophytosis with negative laboratory reports.
Direct microscopy:
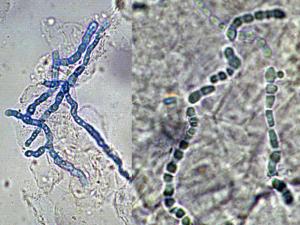
Skin Scrapings, nail scrapings and epilated hairs should be examined using 10% KOH and Parker ink or calcofluor white mounts.
Click images below to expand:

Cultures of T. rubrum.
Culture:
Specimens should be inoculated onto primary isolation media, like Sabouraud's dextrose agar containing cycloheximide (actidione) and incubated at 26-28C for 4 weeks. The growth of any dermatophyte is significant.
Serology:
Not required for diagnosis.Identification:
Characteristic clinical, microscopic and culture features.
See descriptions of individual species for details.Management:
Treatment of dermatophytosis is often dependant on the clinical setting. For instance uncomplicated single cutaneous lesions can be adequately treated with a topical antifungal agent, however topical treatment of scalp and nail infections is often ineffective and systemic therapy is usually needed to cure these conditions. Chronic or widespread dermatophyte infections, acute inflammatory tinea and "Moccasin" or dry type T. rubrum infection involving the sole and dorsum of the foot usually also require systemic therapy. Ideally, mycological confirmation of the clinical diagnosis should be gained before systemic antifungal treatment is commenced.
Further reading:
Elewski, B.E. 1992. Cutaneous fungal infections. Topics in dermatology. Igaku-Shoin, New York and Tokyo.
Hay, R.J. 1992. Clinical manifestations and management of superficial fungal infection in the compromised patient. In Fungal infection in the compromised patient, edited by D.W. Warnock and M.D. Richardson. John Wiley & Sons.
Kwon-Chung, K.J. and J.E. Bennett. 1992. Medical Mycology. Lea & Febiger, Philadelphia and London.
MacKenzie, D.W.R., W. Loeffler, A. Mantovani and T. Fujikura. 1986. Guidelines for the diagnosis, prevention and control of dermatophytosis in man and animals. World Health Organisation WHO/CDS/VPH/86.67. Geneva, Switzerland.Richardson, M.D. and D.W. Warnock. 1993. Fungal Infection: Diagnosis and Management. Blackwell Scientific Publications, London.Rippon, J.W. 1988. Medical Mycology. 3rd Edition. W.B. Saunders Co., Philadelphia, USA. -
Candidiasis - Thrush
Candidiasis is a primary or secondary mycotic infection caused by members of the genus Candida and other related genera. The clinical manifestations may be acute, subacute or chronic to episodic. Involvement may be localized to the mouth, throat, skin, scalp, vagina, fingers, nails, bronchi, lungs, or the gastrointestinal tract, or become systemic as in septicemia, endocarditis and meningitis. In healthy individuals, Candida infections are usually due to impaired epithelial barrier functions and occur in all age groups, but are most common in the newborn and the elderly. They usually remain superficial and respond readily to treatment. Systemic candidiasis is usually seen in patients with cell-mediated immune deficiency, and those receiving aggressive cancer treatment, immunosuppression, or transplantation therapy.
Clinical manifestations:
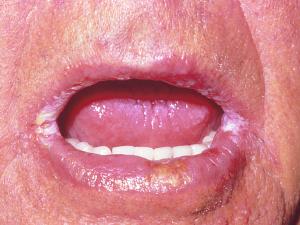
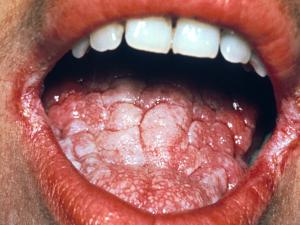
Oropharyngeal candidiasis: including thrush, glossitis, stomatitis and angular cheilitis.
Acute oral candidiasis is rarely seen in healthy adults but may occur in up to 5% of newborn infants and 10% of the elderly. However, it is often associated with severe immunological impairment due to diabetes mellitus, leukemia, lymphoma, malignancy, neutropenia and HIV infection where it presents as a predictor of clinical progression to AIDS. The use of broad-spectrum antibiotics, corticosteroids, cytotoxic drugs, and radiation therapy are also predisposing factors. Clinically, white plaques that resemble milk curd form on the buccal mucosa and less commonly on the tongue, gums, the palate or the pharynx. Symptoms may be absent or include burning or dryness of the mouth, loss of taste, and pain on swallowing.
Click images below to expand:
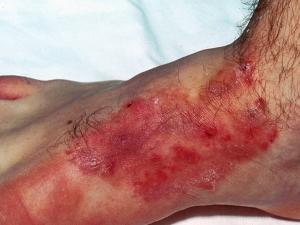
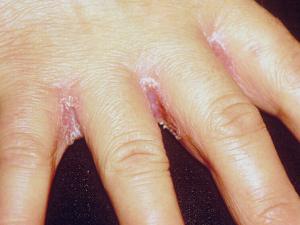
Cutaneous candidiasis: including intertrigo, diaper candidiasis, paronychia and onychomycosis.
Intertriginous candidiasis is most commonly seen in the axillae, groin, inter- and sub-mammary folds, intergluteal folds, interdigital spaces, and umbilicus. Moisture, heat, friction and maceration of the skin are the principle predisposing factors in the normal patient, however obesity, diabetes mellitus, warm water immersion or occlusion of the skin and the use of broad-spectrum antibiotics are additional factors. Lesions consist of a moist, macular erythematous rash with typical satellite lesions present on the surrounding healthy skin.
Click images below to expand:

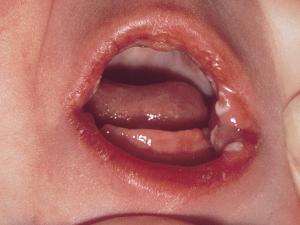
Nappy rash candidiasis in an infant.
Diaper candidiasis is common in infants under unhygienic conditions of chronic moisture and local skin maceration associated with ammonitic irritation due to irregularly changed unclean diapers. Once again characteristic erythematous lesions with erosions and satellite pustules are produced, with prominent involvement of the skin folds and creases.
Paronychia of the finger nails may develop in persons whose hands are subject to continuous wetting, especially with sugar solutions or contact with flour, that macerates the nail folds and cuticle. Lesions are characterized by the development of a painful, erythematous swelling about the affected nails. In chronic cases the infection may progress to cause onychomycosis with total detachment of the cuticle from the nail plate.

Candida onychomycosis showing complete destruction of nail tissue.
Chronic Candida onychomycosis often causes complete destruction of nail tissue and is seen in patients with chronic mucocutaneous candidiasis or other underlying factors that affect either the hormonal or immunologic status of the host. These include diabetes mellitus, hypoparathyroidism, Addison's disease, dysfunction of the thyroid, malnutrition, malabsorption and various malignancies. The use of steroids, antibiotics and antimitotics may also be contributing factors.
Vulvovaginal candidiasis and balanitis:
Vulvovaginal candidiasis is a common condition in women, often associated with the use of broad-spectrum antibiotics, the third trimester of pregnancy, low vaginal pH and diabetes mellitus. Sexual activity and oral contraception may also be contributing factors and infections may extend to include the perineum, the vulva and the entire inguinal area. Chronic refractory vaginal candidiasis, associated with oral candidiasis, may also be a presentation of HIV infection or AIDS. Symptoms include intense vulval pruritus, burning, erythema and dyspareunia associated with a creamy white, curd-like discharge.
In cases of balanitis, diabetes mellitus should be excluded and the sexual partner should be investigated for vulvovaginitis. The symptoms include erythema, pruritus and vesiculopustules on the glan penis or prepuce. Infections are more commonly seen in uncircumcised men and poor hygiene may also be a contributing factor.
Chronic mucocutaneous candidiasis:
Chronic mucocutaneous candidiasis is a form of persistent candidiasis, usually caused by C. albicans, of the skin, nails and mucous membranes that occurs in patients with various metabolic disturbances to cell-mediated immunity. These include defects in leukocyte function or endocrine disorders such as hypoparathyroidism, Addison's disease, hypothyroidism, diabetes, dysfunction of the thyroid and polyglandular autoimmune disease. The patients are usually children. Candida granuloma is a severe localized form which may occur with or without endocinopathy characterized by marked hyperkeratic granulomatous lesions.
Neonatal and congenital candidiasis:
Low birthweight and age, prolonged intravascular catheterization and the use of antibiotic drugs are the principle predisposing conditions for systemic candidiasis in neonates. Blood cultures are often positive and there is also a high incidence of meningitis. Renal complications due to fungus ball formation in the ureters or renal pelvis may also occur. Congenital candidiasis acquired in utero is usually confined to the skin in the form of a generalized erythematous vesicular rash, however intrauterine candidiasis may also result in abortion.
Oesophageal candidiasis:
Oesophageal candidiasis is frequently associated with AIDS and severe immunosuppression following treatment for leukemia or solid tumors. Concomitant oral candidiasis is often present. Oesophagitis may also lead to septicemia and disseminated candidiasis. Symptoms include burning pain in the substernal area, dysphagia, nausea and vomiting. The clinical diagnosis relies on radiological and endoscopic findings, which usually shows white mucosal plaques with erythema resembling those seen in oral candidiasis. Herpes simplex or cytomegalovirus (CMV) infection may also be present and the clinical diagnosis may need to be confirmed by histopathology and culture.
Gastrointestinal candidiasis:
Patients with acute leukemia or other hematological malignancies may have numerous ulcerations of the stomach and less commonly the duodenum and intestine. Perforation can lead to peritonitis and hematogenous spread to the liver, spleen and other organs. Colonization and invasion of the stomach or intestinal mucosa is often accompanied by the excretion of large numbers of yeasts which may be detected in stools.
Pulmonary candidiasis:
Pulmonary candidiasis can be acquired by either hematogenous dissemination causing a diffuse pneumonia or by bronchial extension in patients with oropharyngeal candidiasis. Aspiration of yeasts from the oral cavity has also been reported in infants. Pulmonary candidiasis is difficult to diagnose due to non-specific radiological and culture findings and most patients, especially those with granulocytopenia, present at autopsy. The presence of yeasts in alveolar lavage or sputum specimens is not specific and blood cultures may also be negative. Unfortunately, only histopathology can provide a definitive diagnosis and this is not always possible in patients with coagulation problems.
Peritonitis:
Candida peritonitis can result from colonization of indwelling catheters used for peritoneal dialysis (CAPD) or gastrointestinal perforation due to ulcers, diverticular colitis, surgery or intra-abdominal neoplasm. Symptoms include fever, abdominal pain, tenderness and a cloudy peritoneal dialysate containing greater than 100 leukocytes/mm3. Candida peritonitis usually remains localized to the abdominal cavity unless patients are severely immunosuppressed.
Urinary tract candidiasis:
Transient asymptomatic candiduria may occur during antibiotic or corticosteroid treatment which promotes the growth of Candida, throughout the gastrointestinal and genital tracts, and most lower urinary tract infections result from local spread of yeasts from these sites. This condition is most common in women. Candida cystitis or bladder colonization may be caused by prolonged catheterization with concomitant antibiotic treatment, diabetes and glycosuria, anatomical uropathy, previous bladder endoscopy or surgery, diabetic neurogenic bladder, chronic outlet obstruction from prostatic hypertrophy, or pelvic irradiation for cervical cancer.
Renal candidiasis (pyelonephritis) is usually the result of either an ascending infection or more frequently, hematogenous dissemination from another organ focus. Symptoms include fever, rigors, lumbar pain and abdominal pain. The development of a fungus ball in the renal pelvis, although rare may complicate the infection. Predisposing factors for this include constriction of the urinary tract, localized papillary necrosis, urethral or bladder catheters and diabetes. Even though, up to 80% of patients with disseminated candidiasis also have renal infection and associated candiduria, urine cultures alone are not a reliable method for diagnosis of disseminated infection.
The practical problem in a patient with candiduria is to distinguish between colonization and/or contamination and infection. Therefore, it is important to determine whether renal function is present or whether infection is confined to the bladder. Mycological findings are usually inconclusive which makes the clinical parameters important. The following criteria are suggestive of renal infection; the isolation of yeasts in urine specimens obtained by suprapubic aspiration, positive blood cultures and a positive immunodiffusion precipitin test result or serological conversion in a patient with iatrogenic predisposing factors and/or an underlying illness.
It should be noted that many clinicians do not recommend suprapubic aspirates as they are invasive and require additional expertise, especially in immunocompromised patients. Laboratories are also advised on the need to report the isolation of any yeasts from urine specimens obtained from high risk immunosuppressed patients.
Meningitis:
Candida meningitis is a rare entity, predominantly seen in low birthweight neonates with septicemia and in patients with hematological malignancies, complicated neurosurgery or intracerebral prosthetic devices such as ventriculoperitoneal shunts. Symptoms include a feverish meningeal irritation. Diagnosis in the neonate requires a high index of suspicion by the clinician to the possibility of meningitis as a sequel to septicemia. The detection of Candida cells in smears and its isolation from CSF is often difficult.
Hepatic and hepatosplenic candidiasis:
Hepatosplenic candidiasis occurs in patients with severe neutropenia, usually acute leukemia. Symptoms include fever, hepatosplenomegaly and increased blood concentrations of alkaline phosphatases. Histopathology shows diffuse hepatic and/or splenic necrotic lesions or abscesses containing small numbers of pseudohyphae. However, blood and biopsy cultures are usually culture negative. A definitive diagnosis is often difficult due to the inability to adequately biopsy these patients.
Endocarditis, myocarditis and pericarditis:
Endocarditis is the most common form of cardiac candidiasis. Pre-existing valvular disease with concomitant intravenous catheterization and antibiotic treatment, intravenous drug abuse, heart surgery and valve prosthesis are the most common predisposing factors. Clinical symptoms include fever, murmur, congestive heart failure, anemia and splenomegaly. Blood cultures are often positive and echocardiology and serology for the detection of Candida antibodies (immunodiffusion precipitin tests) are other useful diagnostic procedures. Myocardial abscesses, arterial emboli and purulent pericarditis are additional rare complications of Candida septicemia or surgery.
Candidemia (Candida septicemia) and disseminated candidiasis:
Candidemia has been defined as the presence of yeasts in the blood with or without visceral involvement. Hematogenous dissemination may then occur to one or more other organ systems with the formation of numerous microabscesses. Candida species have been reported to cause up to 15% of cases of septicemia seen in hospital patients.
Predisposing factors include intravenous catheters, use of antibacterial drugs, urinary catheters, surgical procedures, corticosteroid therapy, neutropenia, severe burns, parental nutrition, and chemotherapy induced impairment of oropharyngeal or gastrointestinal mucosa. A characteristic presentation is antibiotic resistant fevers in the neutropenic patient with tachycardia and dyspnea. Hypotension is also common and skin lesions may also occur.
When yeasts are isolated from blood or from tissue biopsies a diagnosis is straightforward, however this is not often the case. Blood cultures often remain negative even in patients dying from proven disseminated candidiasis, especially in the granulocytopenic patient. If at all possible, suspected foci should be aspirated, including articular, peritoneal, CSF, or even vitreal specimens; and liver and/or lung biopsies should also be performed. However histopathology is more often not a viable option because biopsies are contraindicated due to the patients underlying illness. Finally, the detection of yeasts from more accessible no-sterile sites, like urine, is too common to be of diagnostic value. In this situation, of clinically suspected unproven disseminated candidiasis, only cutaneous and/or ocular lesions can rapidly confirm the diagnosis. Specific, reliable serological tests are still not generally available. Empiric antifungal treatment is usually initiated in these cases.

Endophthalmitis due to Candida.
Ocular candidiasis:
Candida endophthalmitis is often associated with candidemia, indwelling catheters or drug abuse, however it is rare in patients with severe neutropenia. Lesions are often localized near the macula and patients complain of cloudy vision. Exogenous Candida endophthalmitis is rare, but cases have been reported following ocular trauma or surgery. Similarly, conjunctival and corneal infections have also been recorded following trauma.
Osteoarticular candidiasis:
Arthritis may be a late sequel of candidemia in neonates or neutropenic patients. Prosthetic or rheumatoid joints are also prone to infection by Candida either by hematogenous spread or direct inoculation during surgery or intra-articular corticosteroid injection. The knee is the main site involved with pain on weight bearing or on full extension. The diagnosis depends on the isolation of yeasts from joint fluid obtained by needle aspiration or from synovial biopsies.
Other forms of candidiasis:
As candidiasis is an iatrogenic, nosocomial infection which is usually endogenous in origin many other clinical manifestations may occur, especially in the debilitated patient. For example, the reported cutaneous, ocular and arthritic manifestations reported in heroin addicts; fever, rash and myalgia associated with leukemia patients; Candida cholecystitis; Candida prostatitis; pancreatic abscesses; epiglottitis and osteomyelitis, to name a few.
Summary of clinical groups and/or predisposing factors for invasive candidiasis.
Neutropenia (especially >7 days).
Hematological malignancy.
Solid tumor malignancy.
Postsurgical intensive care patients.
Prolonged intravenous catheterization.
Broad-spectrum or multiple antibiotic therapy.
Diabetes mellitis.
Parental nutrition.
Severe burns.
Neonates.
Corticosteroid therapy.
Intravenous drug abuse.Laboratory diagnosis:
Clinical material:
Skin and nail scrapings; urine, sputum and bronchial washings; cerebrospinal fluid, pleural fluid and blood; tissue biopsies from various visceral organs and indwelling catheter tips.
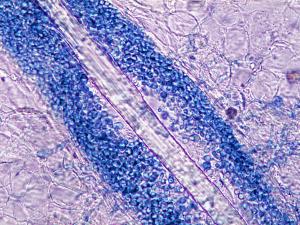
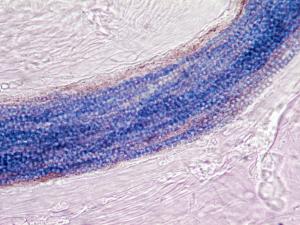
10% KOH mount showing the presence of budding yeast cells and pseudohyphae in a skin scraping and a PAS stained smear showing the presence of budding yeast cells and pseudohyphae in a urine specimen.
Direct microscopy:
(a) Skin and nails should be examined using 10% KOH and Parker ink or calcofluor white mounts; (b) Exudates and body fluids should be centrifuged and the sediment examined using either 10% KOH and Parker ink or calcofluor white mounts and/or gram stained smears; (c) Tissue sections should be stained using PAS digest, Grocott's methenamine silver (GMS) or Gram stain. Note Candida may be missed in H&E stained sections. Examine specimens for the presence of small, round to oval, thin-walled, clusters of budding yeast cells (blastoconidia) and branching pseudohyphae. Candida pseudohyphae may be difficult to distinguish from Aspergillus hyphae when blastoconidia are not observed as often happens in liver biopsies.Interpretation:
As a rule, a positive direct microscopy from a sterile site, especially a tissue biopsy, should be considered significant, even if the laboratory is unable to culture the yeast. Further, the demonstration of pseudohyphae in scrapings or smears from cutaneous, oral, esophageal and vaginal lesions should be considered significant, provided the clinical manifestations support the diagnosis. However, the finding of just budding yeast cells in such material is of little diagnostic importance. Note, pseudohyphae will not be observed in smears when C. glabrata is involved and the diagnosis will require additional supporting evidence. Direct microscopy of sterile body fluids, such as CSF, vitreous humor, joint fluid and peritoneal fluid is relatively insensitive and positive culture will usually be required to make a diagnosis.
Typical moist colonies and budding yeast cells of Candida.
Culture:
Colonies are typically white to cream colored with a smooth, glabrous to waxy surface.Interpretation:
A positive culture from blood, or other sterile body fluid, or tissue biopsy should be considered significant. Lysis centrifugation is currently the most sensitive method for the isolation of Candida from blood. However, positive culture from non-sterile specimens such as sputum, bronchial lavage, esophageal brushings, urine, stool, and surgical drains are of little diagnostic value. Similarly, culture of skin or mucous membrane lesions without supporting evidence from direct microscopy is not diagnostic. Candida species are commonly isolated from the mouth, vagina, anus, and less often, moist skin surfaces of normal individuals who do not have candidiasis.Serology:
Various serological procedures have been devised to detect the presence of Candida antibodies, ranging from immunodiffusion to more sensitive tests such as counter immunoelectrophoresis (CIE), enzyme-linked immunosorbent assay (ELISA), and radioimmunoassay (RIA). However, these are often negative in the immunocompromised patient, especially at the beginning of an infection. The production of four or more precipitin lines in CIE tests has been reported to be diagnostic of candidiasis in the predisposed patient .Tests for circulating antigen by immunological or non-immunological means have also been developed. Of the non-immunological techniques, use of gas liquid chromography (GLC) to detect mannose derivatives of the cell wall or a metabolic by-product, D-arabinitol, have proved the most useful. The detection of antigen by immunological methods such as ELISA or RIA have been used, however for the small laboratory latex agglutination tests for glycoprotien antigen have proved to be the most useful, although variable results have been reported.
It must be stressed that the interpretation of serological tests for Candida, especially in the neutropenic patient, is often difficult and must be correlated with other diagnostic methods. False-negatives and false-positive results do occur. Hopwood and Evans (1991) provide an excellent review of the current serological methods available.
Identification:
The genus Candida is characterized by globose to elongate yeast-like cells or blastoconidia that reproduce by multilateral budding. Most Candida species are also characterized by the presence of well developed pseudohyphae, however this characteristic may be absent, especially in those species formally included in the genus Torulopsis. Arthroconidia, ballistoconidia and colony pigmentation are always absent. Within the genus Candida, fermentation, nitrate assimilation and inositol assimilation may be present or absent, however, all inositol positive strains produce pseudohyphae.Causative agents:
Candida albicans
Candida catenulata
Candida dubliniensis
Candida glabrata complex
Candida haemulonii
Candida inconspicua
Candida parapsilosis complex
Candida rugosa
Candida tropicalis
Clavispora lusitaniae (formerly Candida lusitaniae)
Cyberlindnera fabianii (formerly Candida fabianii)
Debaryomyces hansenii (formerly Candida famata)
Kluyveromyces marxianus (formerly Candida kefyr)
Meyerozyma guilliermondii (formerly Candida guilliermondii)
Nakaseomyces glabratus (formerly Candida glabrata)
Nakaseomyces nivariensis (formerly Candida nivariensis)
Nakaseomyces bracarensis (formerly Candida bracarensis)
Pichia kudriavzevii (formerly Candida krusei)
Pichia norvegensis (formerly Candida norvegensis)
Torulaspora delbrueckii (formerly Candida colliculosa)
Wickerhamomyces anomalus (formerly Candida pelliculosa)
Yarrowia lipolytica (formerly Candida lipolytica)Management: see treatment guidelines section.
Further reading:
Ajello L and R.J. Hay. 1997. Medical Mycology Vol 4 Topley & Wilson's Microbiology and Infectious Infections. 9th Edition, Arnold London.
Barnett et al. 1990. Yeasts characteristics and identification. Cambridge University Press, New York. Computerised identification key also available.
Chandler FW., W. Kaplan and L. Ajello. 1980. A colour atlas and textbook of the histopathology of mycotic diseases. Wolfe Medical Publications Ltd. London.
Elewski BE. 1992. Cutaneous fungal infections. Topics in dermatology. Igaku-Shoin, New York and Tokyo.
Ellis, D.H., D, Marriott and T. Sorrell. Candidial and Cryptococcal infections. An interactive CD-ROM, Pfizer Australia.
Kreger-van Rij. 1996. [4th edition due out any time] The yeasts a taxonomic study. Elsevier Science Publishes B.V. Amsterdam.
Kwon-Chung KJ and JE Bennett 1992. Medical Mycology Lea & Febiger.
Odds, F.C. 1988. Candida and candidosis. 2nd Ed. Bailliere Tindall, London.
Richardson MD and DW Warnock. 1993. Fungal Infection: Diagnosis and Management. Blackwell Scientific Publications, London.
Rippon JW. 1988. Medical Mycology WB Saunders Co.
Warnock DW and MD Richardson. 1991. Fungal infection in the compromised patient. 2nd edition. John Wiley & Sons